Volume 33, Nº 4, July and August 2020
DOI: https://doi.org/10.36660/ijcs.20200033
REVIEW ARTICLE
The Role of 18F-FDG PET/CT in Cardiac Sarcoidosis
Christiane Wiefels
Olabimpe Lamai
Riina Kandolin
David Birnie
Eugene Leung
Claudio Tinoco Mesquita
Rob Beanlands
Dr. Christiane Wiefels
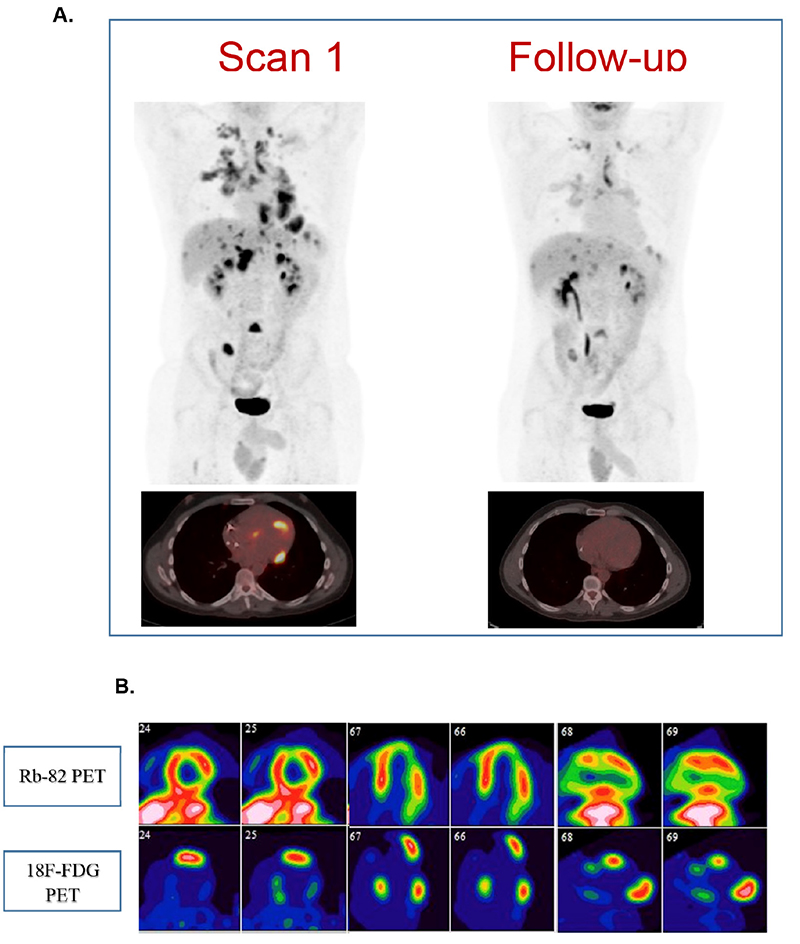
Figure 3 – A. 18F-fluorodeoxyglucose-positron emission tomography (18F-FDG PET) whole body and fused PET/Computed tomography of the heart at baseline and follow-up of a 39-year-old male presenting with episodic complete heart block and sinus node dysfunction. The patient had dual chamber cardioverter defibrillator implantation and was diagnosed with cardiac and systemic sarcoidosis. Treatment with low-dose prednisone and methotrexate was initiated. Scan 1. Whole-body findings: cluster of FDG-avid, tiny nodules in the right lung (SUVmax=6.94), bilateral hilar adenopathy (SUVmax=6.4), bilateral supraclavicular lymph node, multiple foci of increased FDG activity in the liver and spleen and prominent lymph node in the porta hepatis. Cardiac findings: four areas of increased FDG uptake correlating with regions of reduced myocardial perfusion on rest imaging (basal inferolateral wall, basal inferoseptal wall, basal to mid-anterior wall, apical lateral wall and apex). Follow-up of seven months afterwards (Scan 2): Whole-body: Interval reduction in the degree of metabolic activity of all the previous findings. Cardiac findings: no active inflammatory process in the myocardium. Regions of scar in the mid-anterior wall, mid-to- apical inferolateral wall and apex. B. Baseline study with rubidium-82 (top line) and 18F-FDG (bottom line) in short axis, horizontal long axis and vertical long axis, showing the perfusion-metabolism mismatch pattern in the basal inferolateral wall, basal inferoseptal wall, basal to mid-anterior wall, apical lateral wall and apex
Cardiac sarcoidosis (CS) is a rare and potentially fatal condition, characterized by the presence of non-necrotizing granulomatous inflammation and concomitant fibrosis. A variety of clinical manifestation has been described, such as conduction disorders, ventricular arrythmias, congestive heart failure and sudden cardiac death, making a prompt diagnosis and early treatment desirable. Endomyocardial biopsy is the gold-standard diagnostic test, but has a low sensitivity due to the multifocal aspect of the disease. Advanced imaging modalities, such as cardiac magnetic resonance and positron emission tomography (PET) with 18F-fluorodeoxyglucose are now part of the diagnostic criteria and also assist in determining treatment response. However, the interpretation of those studies can be challenging and needs to be made by specialists, as the misdiagnosis could be harmful for the patient. This article describes the pathophysiology of CS and advanced imaging modalities (with a major focus on PET) that should be considered for diagnostic approach and therapy monitoring. Also, newer clinical trials evaluating treatment strategies are described.
Keywords: Cardiomyopathies; Sarcoidosis; Heart Failure; Metabolism Disorders; Sudden Cardiac Death; Diagnostic, Imaging; Magnetic Resonance Imaging/ diagnostic; Positron Emission Tomography Computed Tomography/diagnostic.











